LabWeave
LIMS — Laboratory Information Management System
Custom LIMS solutions built to your SOPs, instrument interfaces, and regulatory requirements — not the other way around.

Laboratory Information Management for Singapore
LIMS That Fits Your Lab — Not a Generic Template
Off-the-shelf LIMS products force laboratories to adapt their standard operating procedures to the software. For research labs, quality assurance teams, and industrial labs with complex or unique workflows, this leads to workarounds, data gaps, and compliance risk.
KEC builds custom LIMS solutions through LabWeave — our own modular laboratory digitalization platform — designed around your exact SOPs, instrument interfaces, sample types, and regulatory framework. Whether you need 21 CFR Part 11 compliance, GxP audit trails, or bespoke integration with your instrument fleet, KEC delivers a system that works the way your lab works.
Lab Challenges We Solve
- Paper-based or spreadsheet-driven workflows with no audit trail
- Manual sample logging causing transcription errors and traceability gaps
- Instrument data not flowing automatically into records
- Compliance reporting (21 CFR Part 11, GxP, ISO 17025) requiring excessive manual effort
- Off-the-shelf LIMS that doesn't match your SOPs or instrument interfaces
- No real-time visibility into sample status or lab throughput

What We Deliver
LIMS Capabilities
Full-stack laboratory digitalization — from sample receipt to regulatory-ready reporting.
Sample Tracking & Chain of Custody
Barcode or RFID-based sample registration, location tracking, and full chain-of-custody logging — from receipt through testing to disposal or archival.
Test Workflow Management
Configurable SOP-driven workflows — assign tests, track progress, capture results, and trigger review/approval gates with role-based access control and e-signatures.
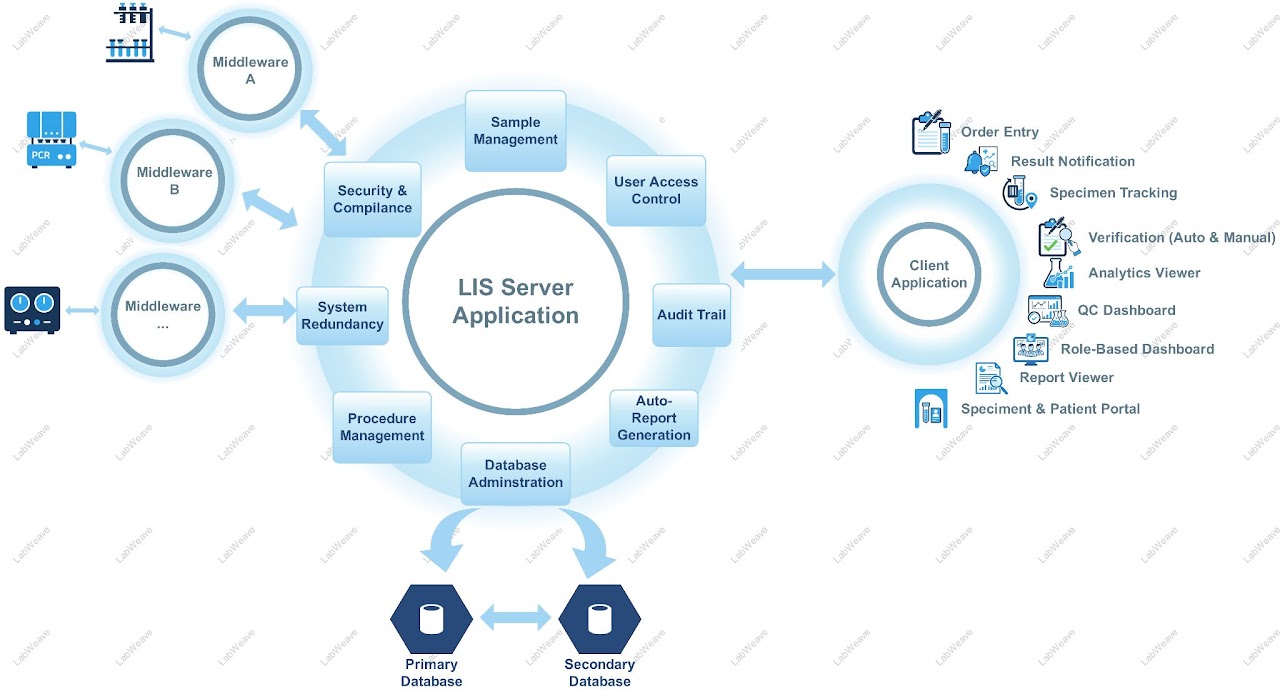
Instrument & System Integration
Bidirectional instrument interfaces — import results directly from analysers, balances, and measurement systems. Integrate with ERP, QMS, and IT/OT platforms.
Compliance & Audit Trail
21 CFR Part 11 e-signature support, immutable audit logs, user action tracking, and automated Certificate of Analysis generation — significantly reducing compliance workload.
LabWeave — A Modular LIMS Built for Flexibility
LabWeave is KEC's own laboratory digitalization platform — modular, fully customisable, and designed to adapt to your unique workflows rather than forcing you to adapt to the software. Purpose-built for Singapore labs that need flexibility without the cost of a full bespoke build.
- Modular architecture — start with what you need, expand later
- Pre-built instrument interfaces for common lab equipment
- Configurable SOP workflows with easy designer interface
- Built-in compliance templates for 21 CFR Part 11 and GxP
- Hosted in Singapore or on-premise deployment options
- Dashboard for real-time sample status and lab throughput monitoring
Core Features
Everything Your Lab Needs
From sample intake to final report — every workflow covered.
Sample Registration & Tracking
Barcode and RFID-based sample intake, unique ID assignment, location tracking, and full chain-of-custody from receipt through testing to archival or disposal.
Result Recording & Report Generation
Capture test results directly from instruments or manual entry, auto-generate Certificates of Analysis, and produce regulatory-ready reports on demand.
Role-Based Access & e-Signatures
Granular user permissions, electronic signatures with audit timestamps, and approval workflow gates — fully aligned with 21 CFR Part 11 requirements.
Real-Time Lab Dashboard
Live visibility into sample queue status, test progress, turnaround times, and lab throughput — giving lab managers the operational oversight they need.
Instrument & ERP Integration
Direct interfaces to lab analysers, balances, and measurement hardware — with bidirectional connectivity to ERP, QMS, and IT/OT enterprise systems.
Data Logging & Audit Trail
Immutable, timestamped records of every system action — meeting GxP, ISO 17025, and 21 CFR Part 11 data integrity requirements without manual effort.
Industry Applications
LIMS Across Singapore's Laboratory Environments
LabWeave adapts to your industry — not the other way around.
Healthcare & Clinical Labs
Patient sample tracking, test workflow automation, result reporting, and full compliance with clinical regulatory standards — all in one configurable platform.
Quality & Manufacturing Labs
Incoming material inspection, in-process testing, and finished product release — with LIMS managing sample queues, instrument results, and QC approval workflows. See our manufacturing work →
Aerospace Materials Testing
Coupon and component test tracking for aerospace qualification programmes — full traceability from material lot to test result to certificate of conformance. See our aerospace work →
R&D & Analytical Laboratories
Flexible LIMS for research labs with evolving test methods — multi-project sample management and integration with data analytics platforms for trend research and deeper insight.